HbA1c
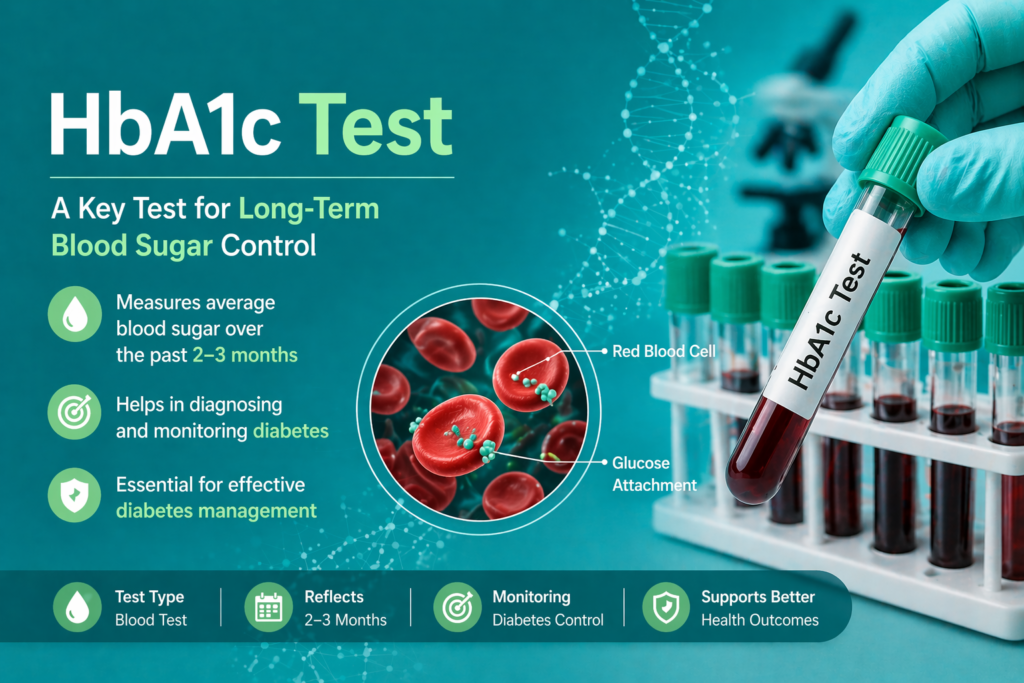
Table of Contents
Definition
HbA1c, or Glycated Hemoglobin, is formed when glucose in the blood binds non-enzymatically to the N-terminal valine of the hemoglobin beta chain. Because red blood cells (RBCs) have an average lifespan of 120 days, the HbA1c level reflects the average blood glucose concentration over the preceding 2–3 months.
Clinical Significance
In the clinical laboratory, HbA1c (Glycated Hemoglobin) is far more than a simple glucose average. Its significance lies in its ability to provide a stable, long-term index of glycemic status, which is unaffected by acute physiological changes like stress, exercise, or recent meals.
Below is a detailed breakdown of its clinical utility for medical lab professionals and clinicians.
1. The “Gold Standard” for Long-term Monitoring
The primary clinical use of HbA1c is to monitor the effectiveness of diabetes therapy.
Glycemic History: While a fasting blood glucose (FBG) test reflects the patient’s status at that specific minute, HbA1c reflects the average blood glucose over the previous 8 to 12 weeks.
Treatment Adjustments: Clinicians use serial HbA1c measurements to determine if a patient’s current regimen (insulin, oral hypoglycemics, or lifestyle changes) is working. A rising HbA1c suggests non-compliance or the need for a dosage increase.
2. Diagnosis and Screening (WHO/ADA Criteria)
Since 2010, the World Health Organization (WHO) and the American Diabetes Association (ADA) have recognized HbA1c as a primary diagnostic tool.
Pre-diabetes Detection: Identifying patients in the 5.7% to 6.4% range allows for early intervention, potentially preventing the progression to full-blown Type 2 Diabetes.
Asymptomatic Screening: Because it doesn’t require fasting, it is the preferred screening tool in emergency departments or during routine check-ups where the patient has already eaten.
3. Predicting Microvascular Complications
The landmark DCCT (Diabetes Control and Complications Trial) and UKPDS (UK Prospective Diabetes Study) established that HbA1c levels are directly proportional to the risk of vascular damage.
Retinopathy: Sustained high HbA1c leads to retinal vessel damage and blindness.
Nephropathy: Chronic hyperglycemia causes glomerular damage. Monitoring HbA1c is critical alongside Urine ACR (Albumin-to-Creatinine Ratio) to prevent end-stage renal disease.
Neuropathy: Nerve damage (leading to “diabetic foot”) is highly correlated with HbA1c levels consistently above 7.0%.
4. Risk Assessment for Macrovascular Disease
Beyond small vessels, HbA1c is an independent risk factor for Cardiovascular Disease (CVD).
Every 1% increase in HbA1c is associated with a significant increase in the risk of myocardial infarction (heart attack) and stroke, even in non-diabetic individuals.
In the lab, a high HbA1c often prompts the clinician to order a Lipid Profile to assess the patient’s overall metabolic cardiovascular risk.
5. Identification of “Stress Hyperglycemia”
In hospital settings (ICU or ER), patients often present with high blood glucose due to the “fight or flight” response (cortisol release).
An HbA1c test can differentiate between acute stress-induced hyperglycemia (where HbA1c will be normal) and undiagnosed chronic diabetes (where HbA1c will be elevated).
6. Surgical and Pregnancy Outcomes
Pre-operative Optimization: Surgeons often require an HbA1c below a certain threshold (e.g., <7.0% or 8.0%) before elective surgery to reduce the risk of post-operative infections and poor wound healing.
Gestational Monitoring: Although not used to diagnose gestational diabetes, it is used to monitor pre-existing diabetics who become pregnant to prevent congenital malformations (teratogenesis) caused by high glucose levels during organogenesis.
Summary Table: HbA1c vs. Fasting Glucose
| Feature | Fasting Plasma Glucose (FPG) | HbA1c |
| Time Period | Current moment | 2–3 months |
| Fasting Required? | Yes (8+ hours) | No |
| Short-term biological variation | High (Stress, illness, meals) | Low (Very stable) |
| Reflects | Acute Glucose Homeostasis | Cumulative Glycemic Burden |
| Primary Use | Acute Diagnosis / Daily monitoring | Long-term Management / Screening |
Methodology & Principle
Measuring HbA1c requires separating the glycated fraction (A1c) from non-glycated adult hemoglobin (A0). Below are the three most clinically significant methodologies used in automated analyzers.
1. Ion-Exchange High-Performance Liquid Chromatography (HPLC)
This is the “reference method” used by most large-scale diagnostic centers.
The Principle: It utilizes the difference in electrical charge between glycated and non-glycated hemoglobin.
The glucose molecule attached to the N-terminal valine of the beta chain reduces the positive charge of hemoglobin.
When the sample is passed through a negatively charged resin (stationary phase), the different hemoglobin fractions (A1a, A1b, F, A1c, and A0) elute at different times based on their specific ionic strengths.
The Process:
Hemolysis: Whole blood is lysed to release hemoglobin.
Injection: The hemolysate is injected into the HPLC column.
Elution: Buffers with increasing ionic strength (mobile phase) push the hemoglobin through the resin.
Detection: A spectrophotometer measures the absorbance at 415 nm.
Quantification: The area under the curve (AUC) for the A1c peak is calculated as a percentage of the total hemoglobin area.
2. Turbidimetric Inhibition Immunoassay (TINIA)
Commonly used on general chemistry analyzers (like Roche Cobas or Abbott Alinity).
The Principle: This method relies on a specific antigen-antibody reaction.
It uses antibodies specifically developed against the first few amino acids of the glycated N-terminal end of the beta-globin chain.
The Process:
Total Hb Measurement: Hemoglobin is converted to a stable derivative and measured colorimetrically.
A1c Measurement: Glycated hemoglobin in the sample binds to anti-HbA1c antibodies.
Agglutination: A “polyhapten” (a reagent containing multiple HbA1c epitopes) is added. It reacts with the excess (unbound) antibodies to form an insoluble complex.
Inverse Relationship: The turbidity (cloudiness) is inversely proportional to the concentration of HbA1c in the patient’s sample. The more HbA1c the patient has, the fewer antibodies are left to react with the polyhapten, resulting in less turbidity.
3. Boronate Affinity Chromatography
Often used in Point-of-Care (POC) devices or for patients with known hemoglobin variants.
The Principle: This method ignores charge and focuses on chemical structure.
It uses the specific affinity of boronic acid for the cis-diol groups of glucose molecules.
The Process:
The resin in the column contains boronic acid.
As the blood passes through, the glycated hemoglobin “sticks” to the boronate, while all non-glycated hemoglobin (A, S, C, F) passes right through.
A second buffer then releases (elutes) the glycated fraction.
Clinical Advantage: Since it only looks for glucose “tags,” it is not affected by common hemoglobin variants (like HbS or HbC trait) that usually interfere with HPLC charge-based separation.
Comparison of Methodologies
| Feature | Ion-Exchange HPLC | Immunoassay | Boronate Affinity |
| Primary Driver | Electrical Charge | Molecular Structure | Chemical Affinity |
| Precision | Excellent (Gold Standard) | High | Good |
| Interferences | Hb Variants (S, C, F) | Minor variant interference | Minimal interference |
| Main Use | High-volume Ref Labs | Routine Chemistry Labs | POC / Variant Testing |
Specimen Requirements
To ensure accurate HbA1c results, the pre-analytical phase is the most critical. As a lab professional, you must ensure the integrity of the red blood cells (RBCs), as any lysis or alteration in the blood-to-anticoagulant ratio can lead to significant diagnostic errors.
Comprehensive Specimen Requirements for HbA1c
1. Sample Type and Collection Container
HbA1c testing requires whole blood to measure the glucose bound within the erythrocytes.
- Preferred Tube: EDTA (Lavender/Purple Top). K2-EDTA (dipotassium) is generally preferred over K3-EDTA in modern vacuum systems due to less cellular shrinkage.
Alternative (Method Dependent): Lithium Heparin (Green Top) may be acceptable for certain immunoassay platforms, but never for HPLC, as heparin can interfere with the chromatographic peaks.
Incompatible Tubes: * Serum (Gold/Red): Clotting destroys the RBCs, making analysis impossible.
Sodium Citrate (Blue): The liquid anticoagulant dilutes the sample, leading to falsely low results.
Fluoride Oxalate (Grey): While used for glucose, the oxalate can interfere with some enzymatic HbA1c methods.
2. Patient Preparation
Fasting: Not required. HbA1c can be drawn at any time of day, regardless of the patient’s last meal.
Hydration: Normal hydration is recommended to ensure easy venous access.
Medication Note: Patients should not stop taking diabetes medications unless directed by a physician, but the lab should be aware of high-dose Vitamin C or E, which can sometimes interfere with certain oxidative methodologies.
3. Collection Technique (The “10-Inversion Rule”)
Because EDTA is a spray-dried additive, immediate and thorough mixing is mandatory.
- Inversions: Gently invert the tube 8–10 times immediately after draw.
Risk of Poor Mixing: Inadequate mixing leads to micro-clots. These clots can clog HPLC capillaries or result in an inhomogeneous sample, causing the analyzer to report a “sampling error” or provide an inaccurate low count.
4. Specimen Volume
Standard Draw: 2.0 mL to 4.0 mL (Standard adult vacuum tube).
Pediatric/Capillary: 250 µL to 500 µL in a microtainer.
Critical Constraint: Underfilling the tube (<50% capacity) increases the EDTA concentration relative to the blood, which can cause RBC shrinkage and potentially affect results on some high-sensitivity platforms.
5. Storage and Stability (2026 Guidelines)
HbA1c is remarkably stable compared to plasma glucose, but temperature control is still vital to prevent RBC degradation.
| Storage Condition | Temperature | Stability Window |
| Room Temperature | 18°C – 25°C | 24 to 72 hours (Analyze promptly) |
| Refrigerated | 2°C – 8°C | 7 to 14 days (Gold Standard for transport) |
| Frozen | -20°C or below | Stable for months (Only for QC/Research) |
Normal Ranges & Clinical Interpretations
In accordance with NGSP (National Glycohemoglobin Standardization Program) and ADA (American Diabetes Association) guidelines, the interpretation of HbA1c is divided into diagnostic categories and therapeutic targets.
1. Diagnostic Thresholds (For Screening & Diagnosis)
These ranges apply to patients not yet diagnosed with diabetes.
| HbA1c Level (%) | Classification | Clinical Interpretation & Action |
| < 5.7% | Normal | Indicates healthy glycemic control. No immediate action required. |
| 5.7% – 6.4% | Pre-diabetes | Indicates a high risk of developing Type 2 Diabetes. Requires lifestyle modification and annual screening. |
| ≥ 6.5% | Diabetes | Confirmed diabetes mellitus. Diagnosis usually requires two abnormal results (e.g., HbA1c and FPG) unless symptoms are present. |
2. Therapeutic Targets (For Known Diabetics)
Once a patient is diagnosed, the goal shifts to management and prevention of complications.
< 7.0% (General Target): The standard goal for most non-pregnant adults. It significantly reduces the risk of microvascular complications (nephropathy and retinopathy).
< 6.5% (Stringent Target): Often recommended for younger patients or those with a short duration of diabetes, provided it can be achieved without significant hypoglycemia.
7.1% – 8.5% (Relaxed Target): Appropriate for elderly patients, those with a history of severe hypoglycemia, or individuals with limited life expectancy/advanced comorbidities.
Detailed Clinical Interpretation of Results
A. High HbA1c Levels (Hyperglycemia)
Consistently elevated HbA1c indicates poor glycemic control over the past 90 days.
Insulin Resistance: Common in Type 2 Diabetes; cells fail to respond to insulin, leaving glucose in the bloodstream for glycation.
Therapeutic Failure: Suggests the current dosage of oral hypoglycemic agents (OHAs) or insulin is insufficient.
Acute Stress/Illness: While HbA1c is stable, prolonged illness or chronic stress can cause a gradual rise in average glucose due to cortisol and adrenaline.
B. Low HbA1c Levels (Hypoglycemia or Altered RBCs)
A very low HbA1c (< 4.0%) is rare and often signals underlying pathology rather than “excellent” glucose control.
Chronic Hypoglycemia: May be seen in insulinoma (insulin-secreting tumor) or over-aggressive diabetes treatment.
Shortened RBC Lifespan: This is the most common lab-related cause. If RBCs only live 60 days instead of 120, there is less time for glucose to bind.
Examples: Hemolytic anemia, blood loss, or recent transfusion.
Critical Interferences: When the Result is a “Lie”
As a Subject Matter Expert in the lab, you must identify discrepancies between HbA1c and clinical presentation.
1. Falsely Increased HbA1c
The result appears higher than the patient’s actual average glucose.
Iron Deficiency Anemia: In iron deficiency, RBC production slows down. The resulting population consists of “older” cells that have been circulating longer, accumulating more glucose.
Splenectomy: The “graveyard” for old RBCs is removed, allowing older cells to stay in circulation longer.
Hyperbilirubinemia: High levels of bilirubin can interfere with certain ion-exchange HPLC methods (appearing as a “ghost” peak).
2. Falsely Decreased HbA1c
The result appears lower than the patient’s actual average glucose.
Hemoglobinopathies (HbS, HbC): Variants can interfere with the elution time in HPLC. If a variant is present, the A1c peak may not be fully captured.
Pregnancy: Increased erythropoiesis (new RBC production) lowers the average cell age.
End-Stage Renal Disease (ESRD): Often involves a combination of shortened RBC survival and erythropoietin therapy, both of which lower HbA1c.
Laboratory “Alert” Values
While “critical values” (like a Potassium of 6.5 mmol/L) require immediate phone calls, extreme HbA1c levels also trigger clinical alerts:
HbA1c > 9.0%: Indicates “Very Poor Control.” High risk for acute complications like DKA (Type 1) or HHS (Type 2).
Sudden Change (> 2.0% shift): A rapid increase or decrease between 3-month checks suggests a major change in health status or medication non-compliance.
Quick Stats
| Feature | Details | Critical Lab Insights |
| Test Type | Clinical Biochemistry / Hemato-Chemistry | A unique hybrid test that measures a chemical reaction (glycation) occurring within a hematological component (RBC). |
| Sample Type | Whole Blood (EDTA) | Must be collected in a Lavender Top tube. Unlike most biochemistry tests, serum/plasma is not used because the target analyte is inside the RBC. |
| Primary Metric | NGSP % and IFCC mmol/mol | NGSP (%) is used for clinical diagnosis (e.g., 6.5%), while IFCC (mmol/mol) is the global scientific standard for higher precision. |
| Fasting Required? | No | One of the few metabolic tests unaffected by recent food intake. It can be drawn at any time, making it ideal for ER and walk-in screenings. |
| Standardization | NGSP & IFCC Traceable | Results are strictly standardized to the DCCT reference to ensure that a 7% result is consistent across every lab globally. |
| Turnaround Time | 15–45 Minutes | High-throughput HPLC analyzers can process samples rapidly, though batching is common in smaller laboratory settings. |
| Biological Half-life | ~120 Days | Based on the average lifespan of an Erythrocyte. The result is weighted; approximately 50% of the HbA1c value is determined by the last 30 days. |
| Critical Values | HbA1c > 9.0% | Results above this threshold are flagged as “Poor Control” and indicate a high risk for long-term microvascular damage. |
| Key Limitation | RBC Turnover Rate | Any condition that changes RBC lifespan (Anemia, Splenectomy, Pregnancy) will invalidate the result, regardless of the patient’s actual glucose levels. |
FAQs
1. Why can’t we use HbA1c to diagnose Type 1 Diabetes in acute cases?
In Type 1 Diabetes, blood glucose levels rise so rapidly that the HbA1c may not have time to reflect the change. Diagnosis in these cases relies on Fasting Plasma Glucose (FPG) or Random Blood Sugar (RBS) accompanied by classic symptoms like polyuria and polydipsia.
2. How often should an HbA1c test be performed?
For patients meeting treatment goals and demonstrating stable glycemic control, twice a year is sufficient. For those whose therapy has changed or who are not meeting targets, it should be performed every 3 months (quarterly). Testing more frequently than every 6 weeks is clinically useless due to the RBC lifespan.
3. What is the “Estimated Average Glucose” (eAG)?
The eAG is a calculated value that translates the HbA1c percentage into the same units patients see on their home glucose monitors (mg/dL or mmol/L).
The Formula: eAG (mg/dL) = 28.7 \times HbA1c – 46.7.
4. Can high Vitamin C intake affect HbA1c results?
Yes. Very high doses of Vitamin C (Ascorbic Acid) can lead to falsely low HbA1c results in certain methodologies because it can interfere with the glycation process or the oxidative reactions used in some immunoassay formats.
5. Why do we see a discrepancy between HbA1c and daily glucose logs?
This is often due to glycemic variability. A patient may have many “highs” and “lows” that average out to a “normal” HbA1c, even though their glucose is unstable. Alternatively, underlying anemia or hemoglobin variants may be providing a false lab reading.
6. Is HbA1c reliable in patients with Chronic Kidney Disease (CKD)?
HbA1c is often falsely low in CKD patients. This occurs because these patients frequently have decreased RBC survival, are treated with Erythropoietin (which increases new, non-glycated RBCs), or have iron deficiency from dialysis. In these cases, Fructosamine may be a better marker.
7. Does Fetal Hemoglobin (HbF) interfere with the test?
Yes, particularly in Ion-Exchange HPLC. High levels of HbF (found in some genetic conditions or during pregnancy) can co-elute with HbA1c, leading to a falsely elevated result. Lab techs must inspect the chromatogram for a prominent HbF peak.
8. Can HbA1c be measured from a finger-stick?
Yes, Point-of-Care (POC) HbA1c devices use capillary blood. While convenient, these must be NGSP-certified to be used for diagnostic purposes; otherwise, they should only be used for monitoring.
9. What is the difference between “Glycated” and “Glycosylated”?
While often used interchangeably, “Glycated” is the chemically correct term. It refers to a non-enzymatic bond between glucose and protein. “Glycosylation” refers to an enzymatic process, which is not what occurs in HbA1c formation.
10. Does a blood transfusion affect the result?
Absolutely. A blood transfusion introduces a large volume of donor RBCs with their own glycation levels. You must wait at least 2 to 3 months after a significant transfusion for the HbA1c to accurately reflect the patient’s own glycemic status.